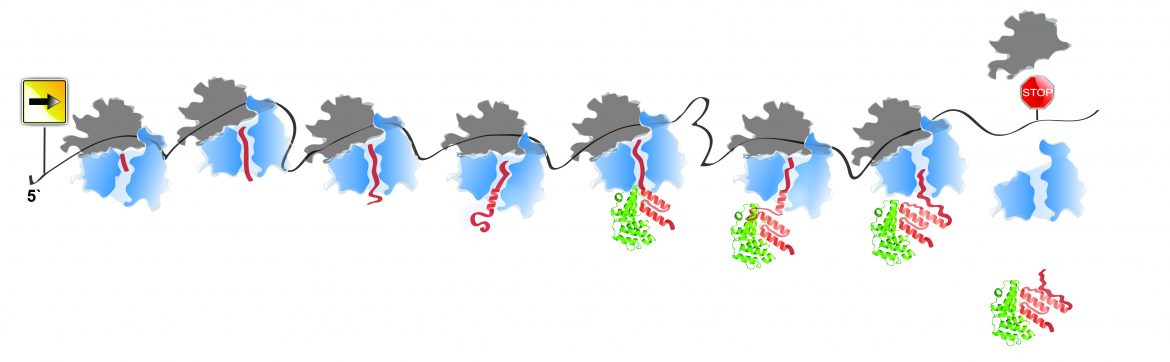
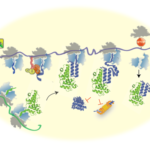
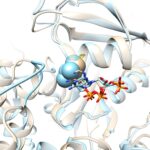
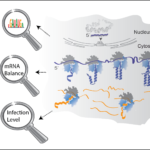
- Assembly
- Chaperones interplay
- in vivo mRNA FISH
- Onset
- Model
- Interface hot spots
- Covid-19 impact on ribosomal function
- Misfolding
The majority of cellular proteins do not function alone; they often act together in complexes to achieve concerted functions.
Despite the prevalence of protein complexes, very little is known of the mechanisms that ensure their correct folding and assembly.
The importance of the folding and assembly challenge is underscored by the growing number of misfolding diseases, such as Alzheimer’s and Parkinson’s. All of which are characterized by the accumulation of misfolded proteins in aggregates.
Here we study how do proteins form, fold and assemble into functional complexes in our cells, what mechanisms facilitate and protect them from aggregation, neurodegeneration, and aging.
Our key questions:
- How are nanomachines formed in our cells?
- How do ribosomes go beyond synthesis to direct the polypeptide-chain folding and assembly?
- How do “lonely” proteins find their complex and pathway partners?
- What co-translational mechanisms protect them from misfolding?
- What co-translational mechanisms degrade “lonely” proteins?
- Do mRNA subcellular organization facilitate complex assembly?
The lab combines a variety of approaches including:
Advanced genetics (Next- Generation Sequencing), Biochemistry and Cell biology, using the awesome power of yeast as a model organism as well as cell culture.
The lab combines a variety of highly advanced methodologies including:
- Selective Ribosome Profiling – capturing the ribosomes engaged by target proteins, in codon resolution
- Single-molecule Fluorescence in situ hybridization (FISH) for imaging mRNAs in the cell
- Developing novel techniques for single-molecule capturing of protein-mRNA interactions